Remoción de Cr (VI) en solución utilizando Theobroma cacao L.: Simulación de columna empacada a escala industrial
Contenido principal del artículo
Resumen
Introducción: Se han desarrollado muchos estudios donde utilizan técnicas para eliminar contaminantes en cuerpos de agua. Entre estas técnicas se encuentra la adsorción, técnica de superficie que utiliza adsorbente a base de residuos agrícolas para remover dichos contaminantes, como los metales pesados pero estos estudios en su mayoría se han realizado a escala laboratorio, son pocas las investigaciones que han buscado anticipar la eficacia del adsorbente a nivel industrial.
Objetivo: Simular la remoción de Cr (VI) en solución mediante una columna empacada a escala industrial utilizando biomasa de Theobroma cacao L. como adsorbente.
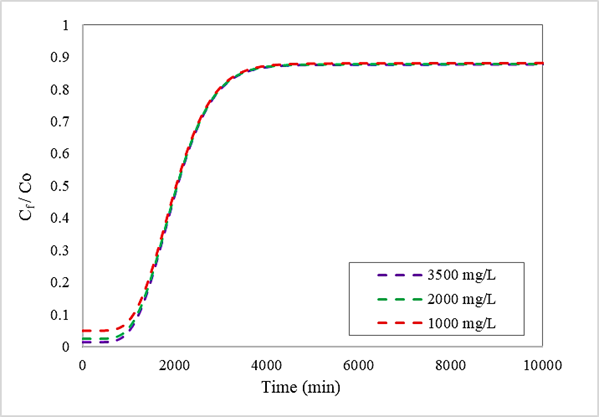
Metodología: Se utilizó un método del tipo de modelado y simulación donde se empleó el instrumento informático Aspen Adsorption, para simular la columna de adsorción empleando diversas configuraciones combinadas con una evaluación paramétrica.
Resultados: Con los descubrimientos realizados se obtuvo que, usando los modelos matemáticos Langmuir y Fuerza Motriz Lineal (LDF) se presentaron eficiencias del proceso de adsorción hasta un 97% de remoción de Cr (VI). Además, los mejores valores paramétricos fueron una altura de la columna de 5m, un caudal de 100 m3/día y una concentración inicial de 3500 mg/L.
Conclusiones: Estos hallazgos permiten presentar este estudio como una forma novedosa en el campo de la ingeniería de cómo las herramientas computacionales tienen la capacidad de predecir el posible comportamiento de columnas de adsorción rellenas con biomasas basadas en residuos orgánicos.
Descargas
Detalles del artículo

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-SinDerivadas 4.0.
Los artículos publicados son de exclusiva responsabilidad de sus autores y no reflejan necesariamente las opiniones del comité editorial.
La Revista INGE CUC respeta los derechos morales de sus autores, los cuales ceden al comité editorial los derechos patrimoniales del material publicado. A su vez, los autores informan que el presente trabajo es inédito y no ha sido publicado anteriormente.
Todos los artículos están bajo una Licencia Creative Commons Atribución-NoComercial-SinDerivadas 4.0 Internacional.