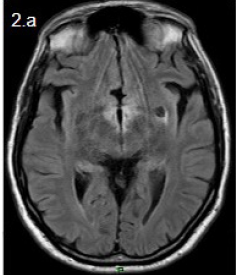
Optic tract and internal capsule lesion in a patient with Wernicke-Korsakoff syndrome
##plugins.themes.bootstrap3.article.main##
Abstract
A 72-year-old man, presented with a one-week history of confusion and an anterograde amnesic disorder accompanied by confabulation, with lack of insight to his symptoms.
Downloads
Download data is not yet available.
##plugins.themes.bootstrap3.article.details##
How to Cite
Hernández, M.-A., Varela, F., & Bensi, C. (2020). Optic tract and internal capsule lesion in a patient with Wernicke-Korsakoff syndrome. Journal of Applied Cognitive Neuroscience, 1(1), 101–103. https://doi.org/10.17981/JACN.1.1.2020.07
Section
Neuroimage

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
You are free to:
- Share — copy and redistribute the material in any medium or format.
- The licensor cannot revoke these freedoms as long as you follow the license terms.
Under the following terms:
- Attribution — You must give appropriate credit, provide a link to the license, and indicate if changes were made. You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
- NonCommercial — You may not use the material for commercial purposes.
- NoDerivatives — If you remix, transform, or build upon the material, you may not distribute the modified material.
- No additional restrictions — You may not apply legal terms or technological measures that legally restrict others from doing anything the license permits.